Next-generationRecombinant Growth FactorsFor Cell Therapy Manufacturing.

Plant-derived raw materials for ex-vivo cell therapy manufacturing.Specifically designed for induced Pluripotent Stem Cells (iPSCs)& Mesenchymal Stem Cells (MSCs) processes.
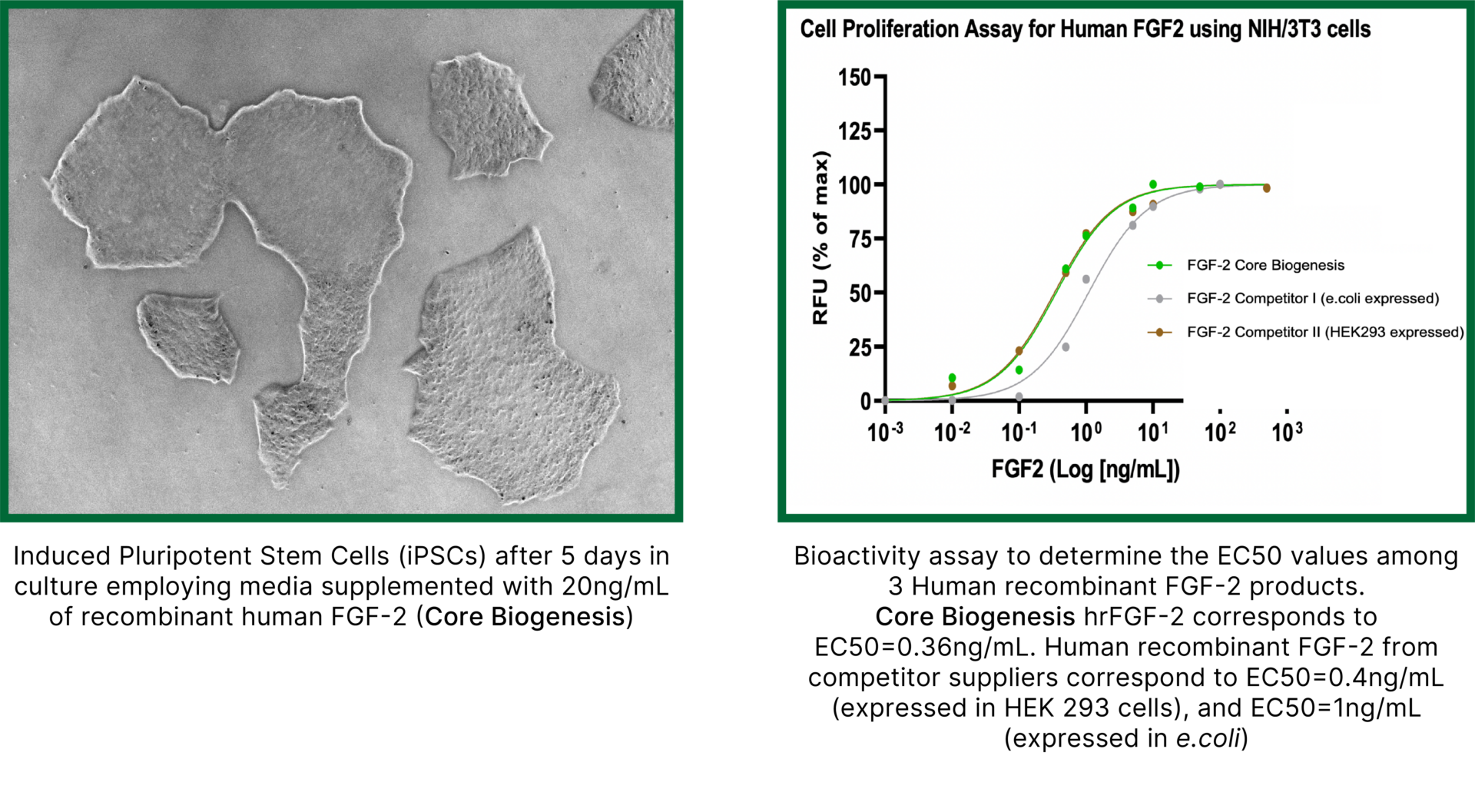
Gold-standard product quality, stability, bioactivity and consistency.
For a seamless transition from the lab to clinics, with the highest levels of quality and safety, our raw materials are manufactured and tested in compliance with:
- ISO 9001 quality management systems standard
- USP Chapter <92>, Growth Factors & Cytokines Used in Cell Therapy Manufacturing
- USP Chapter <1043>, Ancillary Materials for Cell, Gene, and Tissue-Engineered Products
- Ph. Eur. General Chapter 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based Products
Consistency of stem cell cultures is crucial to translate cell therapy and regenerative medicine research toward therapeutic applications. Animal-Free and endotoxin-free recombinant growth factors are critical to reducing variability in Stem Cell cultures. We offer clear raw material qualification, documentation and traceability, and best-in-class lot-to-lot consistency. Our endotoxin-free products drastically reduce lot to lot variability and simplify compliance with regulatory guidelines.
Featured Protein

-High purity & bioactivity
-Animal component-free
-50x longer half-life
- Hassle-free organoid production
-Better cell culture homogeneity
-Weekend-free iPSC culture
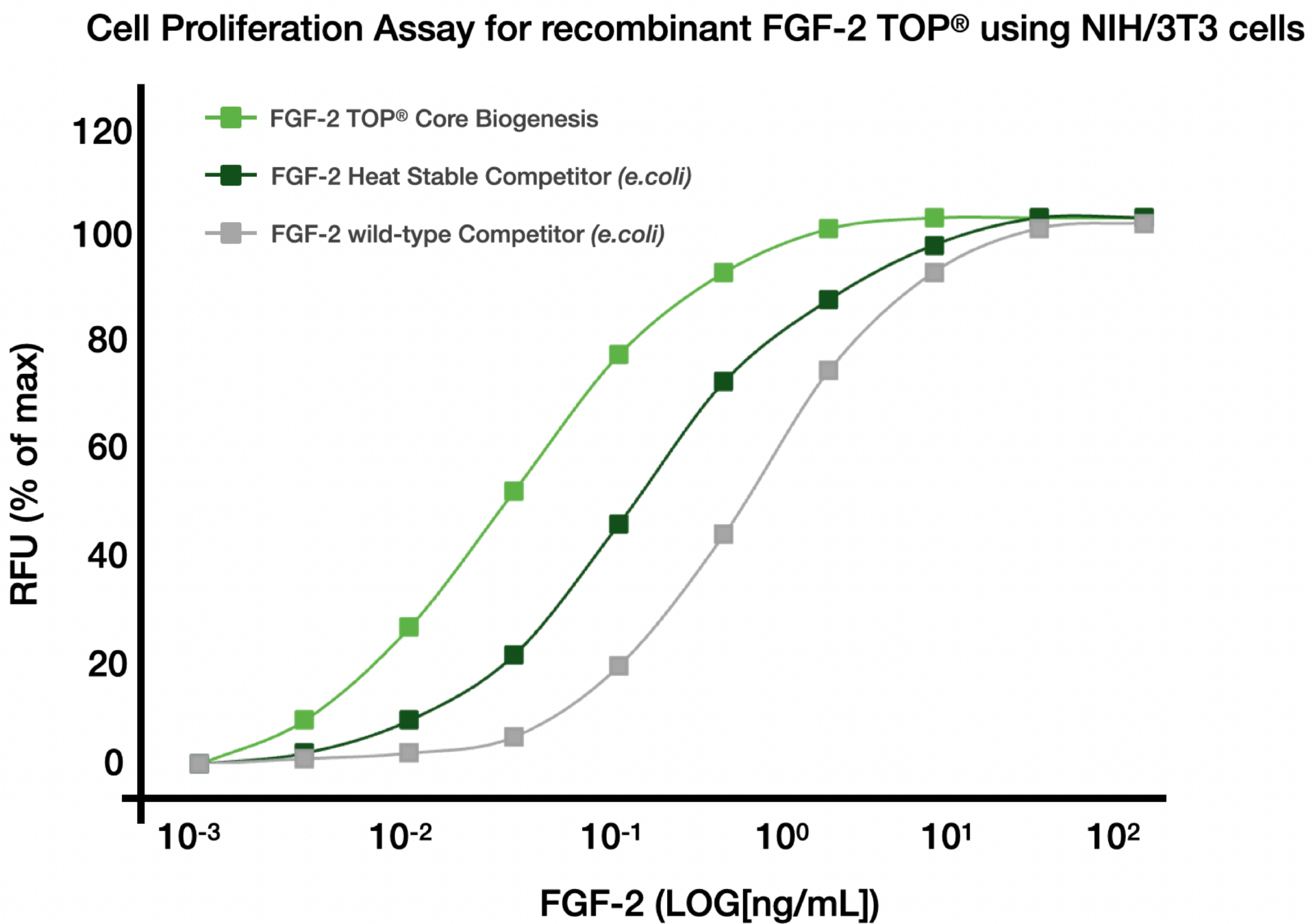
Engineered for heat stability and a 50x longer half-life


References:
Koledova et al. 2019
Fibroblast Growth Factor 2 Protein Stability Provides Decreased Dependence on Heparin for Induction of FGFR Signalling and Alters ERK Signalling Dynamics
Dvorak et al. 2017
Computer-assisted engineering of hyper-stable fibroblast growth factor 2
Improved bioactivity for enhanced results in a variety of applications
References:
Lyra-Leite et.al 2021
An updated protocol for the cost-effective and weekend-free culture of human induced pluripotent stem cells
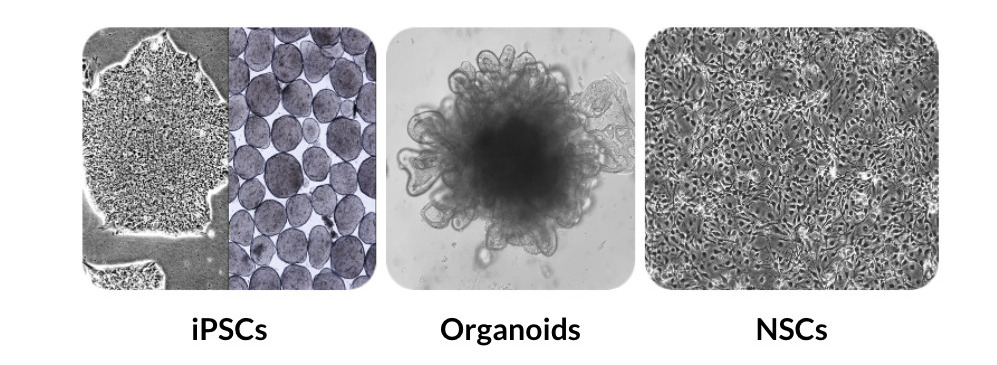
Cellular testing
Cell Characterization employing FGF-2 TOP® (Thermostability Optimized) growth factor in culture and expansion of iPSCs (2D and 3D conditions); Patient-derived Intestinal Organoids; iPSC-derived Neural Stem Cells (NSCs)

